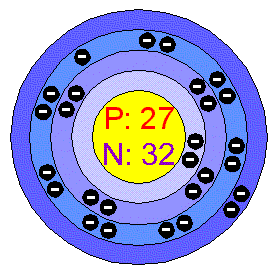 
Correct fitting of multiplet split structure prevents false identification of other chemical states.Cr2p for Cr(III) oxide has many multiplet-split components.A satellite feature of the Cr2p 3/2 peak overlaps the Cr2p 1/2 component in Cr 2O 3.Method for chemical analysis of Cr(III) oxide .How many unpaired electrons would you expect for chromium in Cr (H2O)6 3+ Is this a paramagnetic or diamagnetic material 3. Check for the weaker Te3p peaks, if tellurium concentration is high enough. Write out the entire electron configuration for chromium using spdf notation.Sc3+ has stable electronic configuration (vacant d-orbital), therefore Sc3+ is more stable than. formation, 29-30 cobalt(III) complexes, 2-3, 6, 21, 58-81 absolute configurations. 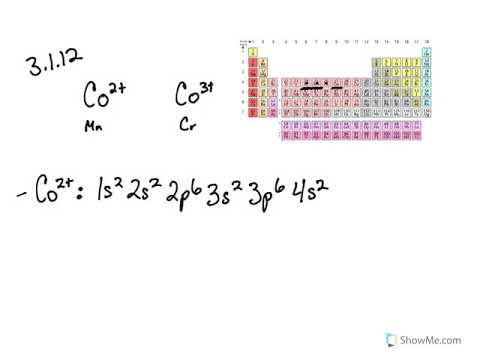
Although Te3d/Cr2p peaks have strong overlap, Te3d spin-orbit components have a 3:2 intensity ratio compared to a 2:1 ratio for Cr2p. Which is a stronger reducing agent Cr2+ or Fe2+ and why. water exchange, 16-17, 19 chromium(III) complexes, 3, 6.the abbreviated electron configurations of the following elements: 6) cobalt. Electron configuration can be done in two ways. In the space below, write the unabbreviated electron configurations of the. Direct overlap between Te3d and Cr2p peaks. The electron configuration of cobalt is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2, if the electron arrangement is through orbitals.Cr2p peak has significantly split spin-orbit components (Δ metal=9.3eV).An ion has the electron configuration 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10. x1 y5 (1) (iii) Annotate the diagram below showing the 4s and 3d orbitals for a chromium atom using an arrow, and, to represent a spinning electron. The Cr atom has 24 electrons the electrons get filled with the lowest energy orbital first and later the remaining. The secondary chromium peak, Cr3p, shows reasonably large chemical shifts and may be easier to peak fit in this situation. of Argon (1) (ii) State the values of x and y. Stellite alloys, which contain cobalt, chromium and tungsten, are used to make high-speed and high temperature.In the presence of high concentrations of zinc, it may be difficult to assign chromium chemistry using the Cr2p region, due to strong overlap with the Zn LMM Auger region.Use weaker Cr3p peak instead if chromium concentration is high enough to give good Cr3p signal.Cr2p overlaps strongly with Zn LMM Auger peaks, e.g., in steel samples.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
